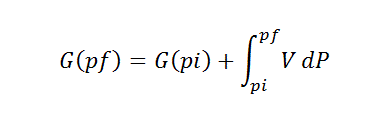Thermodynamic properties of fluids in homogeneous phase in closed systems:
What we have to know...
D stands for delta
>1st law: du=dQ + dW ......... we also know that dW=-PdV
>2nd law: dQ = TdS
So we can assume:
dU= TdS - PdV
What happen when the Pressure or the Temperature vary?
How G varies with P or T?
First we deduce the formula...
1st we have the Gibbs free energy formula:
dG= dH - dTS
From the variables:
dH =dU + dPV = dU + PdV + VdP
dTS= TdS + SdT
Therefore:
dG= dU+ PdV + VdP - TdS -SdT
DG= TDS -pdv +pdv +vdp-tds-sdt
*Color variables are cancelled.
THEREFORE:
DG= VDP -SDT
FUNDAMENTAL EQUATION OF CHEMICAL THERMODYNAMICS.
AND NOW IF THE TEMPERATURE IS CONSTANT, HOW GIBBS ENERGY CHANGES WITH DP?
IF T= CONSTANT, DT=0.
THEREFORE:
DG= VDP
AS WE HAVE THE DIFFERENTIAL OF PRESSURE WE HAVE TO INTEGRATE AS THE FOLLOWING:
DG= VDP -SDT
FUNDAMENTAL EQUATION OF CHEMICAL THERMODYNAMICS.
AND NOW IF THE TEMPERATURE IS CONSTANT, HOW GIBBS ENERGY CHANGES WITH DP?
IF T= CONSTANT, DT=0.
THEREFORE:
DG= VDP
AS WE HAVE THE DIFFERENTIAL OF PRESSURE WE HAVE TO INTEGRATE AS THE FOLLOWING: