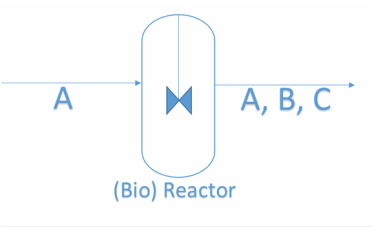
This topic studies strategies to separate a 2-component homogeneous system and estimate the degree to which each specie will transfer.
The big question: How?
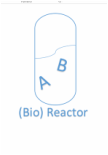
So the big question is how does a counterpart system with two components is separated?
If you start looking, you will notice that among the techniques used to separate compounds from a homogenos system are distillation and crystallization. These two techniques are based on phase separation, the most common method for separating homologous substances, this is to use the different boiling or melting points of a substance to separate it from other causing phase change in one before the other.
If you start looking, you will notice that among the techniques used to separate compounds from a homogenos system are distillation and crystallization. These two techniques are based on phase separation, the most common method for separating homologous substances, this is to use the different boiling or melting points of a substance to separate it from other causing phase change in one before the other.
Raoult's Law
As we know now the separation processes involves phase-separation, this can be achieved in different ways:
P=xP*dis --> Vapor pressure (Ideal solution)
We know that in a phase equilibrium, the fugacity of steam will be equal to the fugacity of the liquid,
- Gas-liquid
- Solid-liquid
- Gas-solid
- Liquid-liquid (inmisable)
P=xP*dis --> Vapor pressure (Ideal solution)
We know that in a phase equilibrium, the fugacity of steam will be equal to the fugacity of the liquid,
Finally joining the expressions of vapor and liquid, our final expression is:
YiP=XiPsat
YiP=XiPsat