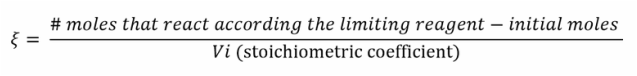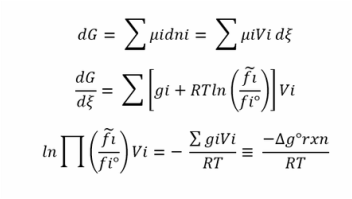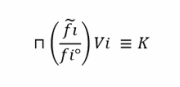Introduction
In this topic we look at the chemical equilibrium and we will study it from the point of view of the mathematical models of thermodynamics.
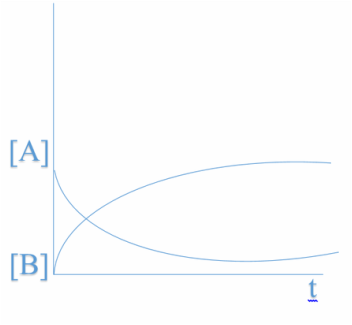
Let's suppose the reaction... A ---------- B+ C When it enters the zone of equilibrium in reaction (when the graphics area both constant concentration of reactants and products is reached), it is when we say that the dynamic equilibrium in the reaction is reached. This means that the speed of reaction of A equals the speed of reaction products B and C. In the equilibrium sA (speed of A) = sB (speed of B) |
Basic concepts
The sA and sB can be calculated as follows...
sA = - (dCA) / dt sB= dCB / dt
where:
d stands for Delta.
CA and CB are the concentrations of A and B respectively
t time of the reaction.
The "-" in dCA is because as A refers to the reactants, as the reaction takes place these are decreasing and conversely products are formed.
The percent of conversion of the reaction can be calculated as ...
%cov = (Mr / Mf) x 100
where:
Mr: Moles reacting
Mf: Feed moles
But... how do we calculate the reacting moles??
The answer is simple, we calculate that with the function of degree of progress:
sA = - (dCA) / dt sB= dCB / dt
where:
d stands for Delta.
CA and CB are the concentrations of A and B respectively
t time of the reaction.
The "-" in dCA is because as A refers to the reactants, as the reaction takes place these are decreasing and conversely products are formed.
The percent of conversion of the reaction can be calculated as ...
%cov = (Mr / Mf) x 100
where:
Mr: Moles reacting
Mf: Feed moles
But... how do we calculate the reacting moles??
The answer is simple, we calculate that with the function of degree of progress:
For the kinetic equation we can calculate:
sA = k[A]
sB= k[B]
And the the rate law goes like this:
r = k[A]^m [B]^n
where:
[A] and [B] are the concentrations of the reactants and products respectively
k: are the constants of speed.
m and n: are the orders of reaction
So then we came to
kA / kB = [B]^n / [A]^m
Kc = [D]^d [C]^c / [A]^a [B]^b --------> THIS IS THE CONSTANT OF EQUILIBRIUM.
sA = k[A]
sB= k[B]
And the the rate law goes like this:
r = k[A]^m [B]^n
where:
[A] and [B] are the concentrations of the reactants and products respectively
k: are the constants of speed.
m and n: are the orders of reaction
So then we came to
kA / kB = [B]^n / [A]^m
Kc = [D]^d [C]^c / [A]^a [B]^b --------> THIS IS THE CONSTANT OF EQUILIBRIUM.
tHERMODynamic equilibrium
What you need to know...
As we remember in equilibrium DG = 0
and ...
DG = DG° + RTlnQ = 0
In equilibrium Q=K and lnK = DG°/-RT
And finally...
K=e^(-DG/RT)
Fow mixtures:
- We can change T (temperature), P (pressure) or C (concentration) in order to change the K
- DH (Delta entalpy) (+) means that the reaction is endothermic so in order to promote the formation of products we need to raise the temperature.
- DH (-) means that the reaction is exothermic so in order to promote the formation of products we need to decrease the temperature.
- Q = Kc means equilibrium
- Q > Kc means that are more products and that the reaction tends to the left
- Q < Kc means that are more reactants and that the reaction tends to the right
- ** Remember that the Q is calculated as Kc but at any time!
As we remember in equilibrium DG = 0
and ...
DG = DG° + RTlnQ = 0
In equilibrium Q=K and lnK = DG°/-RT
And finally...
K=e^(-DG/RT)
Fow mixtures:
So strictly the criterion for chemical equilibrium is given by the following expression