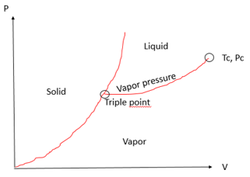
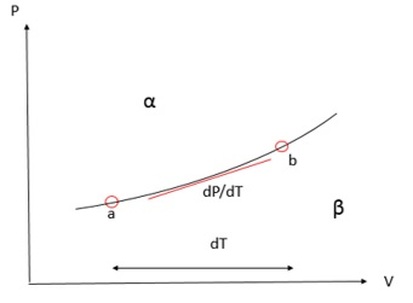
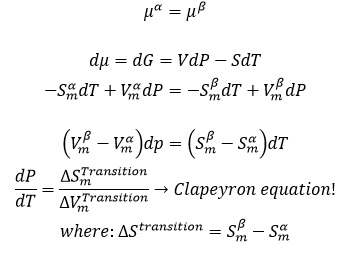Phase Diagram:
Map of P and T at which each phase of a substance is more stable.
Phase:
Form of matter that is uniform throughout in chemical composition and physical state.
Phase transition:
Spontaneous conversion of one phase into another, and occurs at specific T for a given P. Liquid water at 5°C, 1 atm is more stable at T > 0, G < 0.
Homogeneous – 1 phase.
Heterogeneous – 2 or more phases.
Map of P and T at which each phase of a substance is more stable.
Phase:
Form of matter that is uniform throughout in chemical composition and physical state.
Phase transition:
Spontaneous conversion of one phase into another, and occurs at specific T for a given P. Liquid water at 5°C, 1 atm is more stable at T > 0, G < 0.
Homogeneous – 1 phase.
Heterogeneous – 2 or more phases.
State postulate of pure component:
All intensive thermodynamic properties can be determined from two independent intensive properties.
Degrees of Freedom:
Number of independent intensive properties.
Gibbs Phase Rule (F):
F=C-P+2
C - # of components, P - # of Phases
Phase equilibrium (2 phases) for a pure component:
F=1-2+2
F=1
We need an expression relating the P at which 2 phases co-exist with the T of the system. This will help us to predict Psat.
Solid – Liquid Fusion
Liquid – Solid Freezing
Solid – Vapor Sublimation
Vapor – Solid Deposition
Liquid – Vapor Vaporization
Vapor – Liquid Condensation
All intensive thermodynamic properties can be determined from two independent intensive properties.
Degrees of Freedom:
Number of independent intensive properties.
Gibbs Phase Rule (F):
F=C-P+2
C - # of components, P - # of Phases
Phase equilibrium (2 phases) for a pure component:
F=1-2+2
F=1
We need an expression relating the P at which 2 phases co-exist with the T of the system. This will help us to predict Psat.
Solid – Liquid Fusion
Liquid – Solid Freezing
Solid – Vapor Sublimation
Vapor – Solid Deposition
Liquid – Vapor Vaporization
Vapor – Liquid Condensation
Slope of boundary is given by dP/dT. We already know that:
In the following links, you can study the different state boundaries...