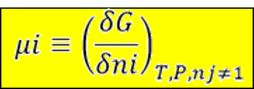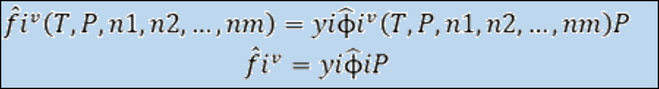IF YOU HAVEN'T SEEN THE PAGE OF EQUILIBRIUM IN MULTICOMPONENT SYSTEMS CLIC IN THE NEXT LINK...
It is the tendency of a component to flow from one phase to another. This happen when one chemical potential is greater than the other. Since any system tends to move towards a state of minimum Gibbs energy and the chemical potential is equal to the partial molar gibbs energy; the chemical potentials will be the same at some point. This means the system has reach equilibrium.
Where:
Criteria for chemical equilibrium in Multicomponent: