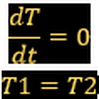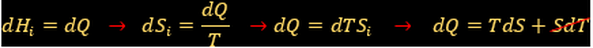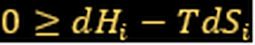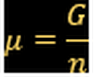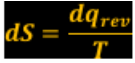
Thermo: heat, energy in transfer
Dynamics: motion
Heat: random motion of the atoms
Work: order motion of atoms
Entropy measures the disorder of the molecules. The disorder tells me the direction of the processes. Measures the energy dispersed in a process. If the total entropy of the system increases, it occurs a spontaneous change.
Steady state: properties don’t change during time.
Dynamics: motion
Heat: random motion of the atoms
Work: order motion of atoms
Entropy measures the disorder of the molecules. The disorder tells me the direction of the processes. Measures the energy dispersed in a process. If the total entropy of the system increases, it occurs a spontaneous change.
Steady state: properties don’t change during time.
Equilibrium: lack of change, absence of ANY tendency to change; a special case of the steady state where it is static. There is no dynamic or heat transferring.
Driving force: any type of influence for a system to change.
Equilibrium: condition in which the state neither changes with time nor has the tendency to spontaneously change. State of maximum total entropy.
Thermodynamic equilibrium: mechanical, thermal and chemical equilibrium simultaneously.
Driving force: any type of influence for a system to change.
Equilibrium: condition in which the state neither changes with time nor has the tendency to spontaneously change. State of maximum total entropy.
Thermodynamic equilibrium: mechanical, thermal and chemical equilibrium simultaneously.
- Thermal equilibrium: Tsys = Tsorr
- Mechanical equilibrium: Psys = Psurround
- Chemical equilibrium:
- Phase equilibrium.- more than one phase with no tendency to change.
- Chemical reaction equilibrium.- when the reaction has no more tendency to react.
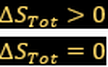
For irreversible change in isolated system:
(Spontaneous change)
(Reversible)
Now, in a closed system with the following assumptions:
· Pure component i
· Mechanical equilibrium (P = cte)
· Thermal equilibrium
· PV work ONLY
· Irreversible process
We have that:
(Spontaneous change)
(Reversible)
Now, in a closed system with the following assumptions:
· Pure component i
· Mechanical equilibrium (P = cte)
· Thermal equilibrium
· PV work ONLY
· Irreversible process
We have that:
Combining 1st and 2nd Law:

New thermodynamic property to determine chemical equilibrium:
Gibbs free energy. Useful energy of the system.
Hi.- is related to useful work
TSi.- is related to energy dissipated
Gi.- Gibbs free energy
If a change is spontaneous Gi < 0, and we talk about an exothermic reaction. If it is non spontaneous Gi > 0, and we have an endothermic reaction. If a system is in equilibrium Gi = 0.
Systems tend to move towards a state of minimum Gibbs energy.
Chemical potencial: A way to calculate Gibbs free energy for one component system.
Gibbs free energy. Useful energy of the system.
Hi.- is related to useful work
TSi.- is related to energy dissipated
Gi.- Gibbs free energy
If a change is spontaneous Gi < 0, and we talk about an exothermic reaction. If it is non spontaneous Gi > 0, and we have an endothermic reaction. If a system is in equilibrium Gi = 0.
Systems tend to move towards a state of minimum Gibbs energy.
Chemical potencial: A way to calculate Gibbs free energy for one component system.
μ.- Chemical potential