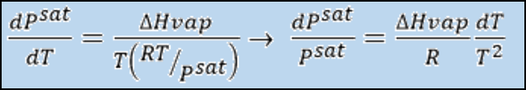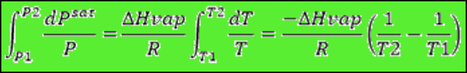IF YOU HAVEN´T SEEN THE PAGE ABOUT THE PHYSICAL TRANSFORMATION OF SUBSTANCES CLIC ON THE NEXT LINK...
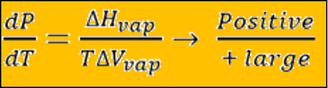
Then Clapeyron equation is:
So: dP/dt is (+) but much smaller than S – L boundary.
Assuming ideal behavior we know that: Vm(g)=RT/P, then the final Clausius Clapeyron Equation would be:
Assuming ideal behavior we know that: Vm(g)=RT/P, then the final Clausius Clapeyron Equation would be:
And if we assume that the change in enthalpy is almost constant: