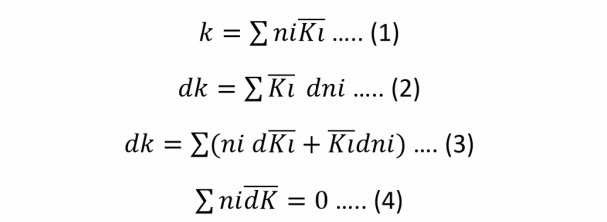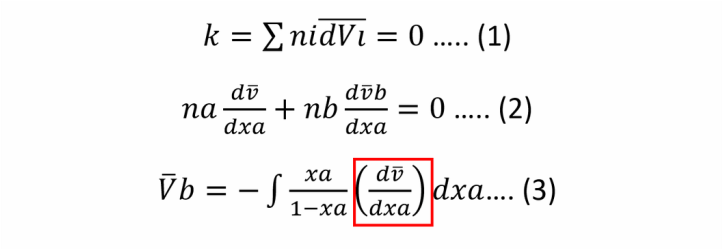IF YOU HAVEN'T SEEN THE PAGE OF EQUILIBRIUM IN MULTICOMPONENT SYSTEMS CLIC IN THE NEXT LINK...
The Gibbs-Dühem Theorem is an equation that gives useful relationship between the partial molar properties of different sepecies in a mixture.
For example:
In a binary mixture (ex: ethanol-water), if you know the value of the molar property of one of the species, using Gibbs-Dühem equation you can calculate the partial molar property value for the other species.
WE DEDUCE THE EQUATION 3 BY DIVIDING BY NTOTAL AND MAKING MATH ARRANGMENT.
THE PART THAT IS IN A RED SQUARE CAN BE CALCULATED AS THE SLOPE IN EXPERIMENTS.
THE PART THAT IS IN A RED SQUARE CAN BE CALCULATED AS THE SLOPE IN EXPERIMENTS.